How To Ideal Gas Law The ideal gas law relates the four independent physical properties of a gas at any time The ideal gas law can be used in stoichiometry problems whose chemical reactions involve gases Standard temperature and pressure STP are a useful set of benchmark conditions to compare other properties of gases At STP gases have a volume of 22 4 L per
The ideal gas law PV nRT 10 3 1 10 3 1 P V n R T relates the pressure volume temperature and number of moles in a gas to each other R is a constant called the gas constant The ideal gas law is what is called an equation of state because it is a complete description of the gas s thermodynamic state At STP gases have a volume of 22 4 L per mole The ideal gas law can be used to determine the density of gases 15 7 Gas Stoichiometry is shared under a CC BY 4 0 license and was authored remixed and or curated by LibreTexts The ideal gas law relates the four independent physical properties of a gas at any time
How To Ideal Gas Law
 How To Ideal Gas Law
How To Ideal Gas Law
https://i.ytimg.com/vi/iaZ96KaQ44c/maxresdefault.jpg
Charles s law states that the volume of a given amount of gas is directly proportional to its temperature on the kelvin scale when the pressure is held constant Mathematically this can be written as V T or V constant T or V k T or V 1 T 1 V 2 T 2
Templates are pre-designed documents or files that can be used for numerous functions. They can conserve time and effort by supplying a ready-made format and design for producing different kinds of material. Templates can be used for personal or professional jobs, such as resumes, invites, leaflets, newsletters, reports, discussions, and more.
How To Ideal Gas Law
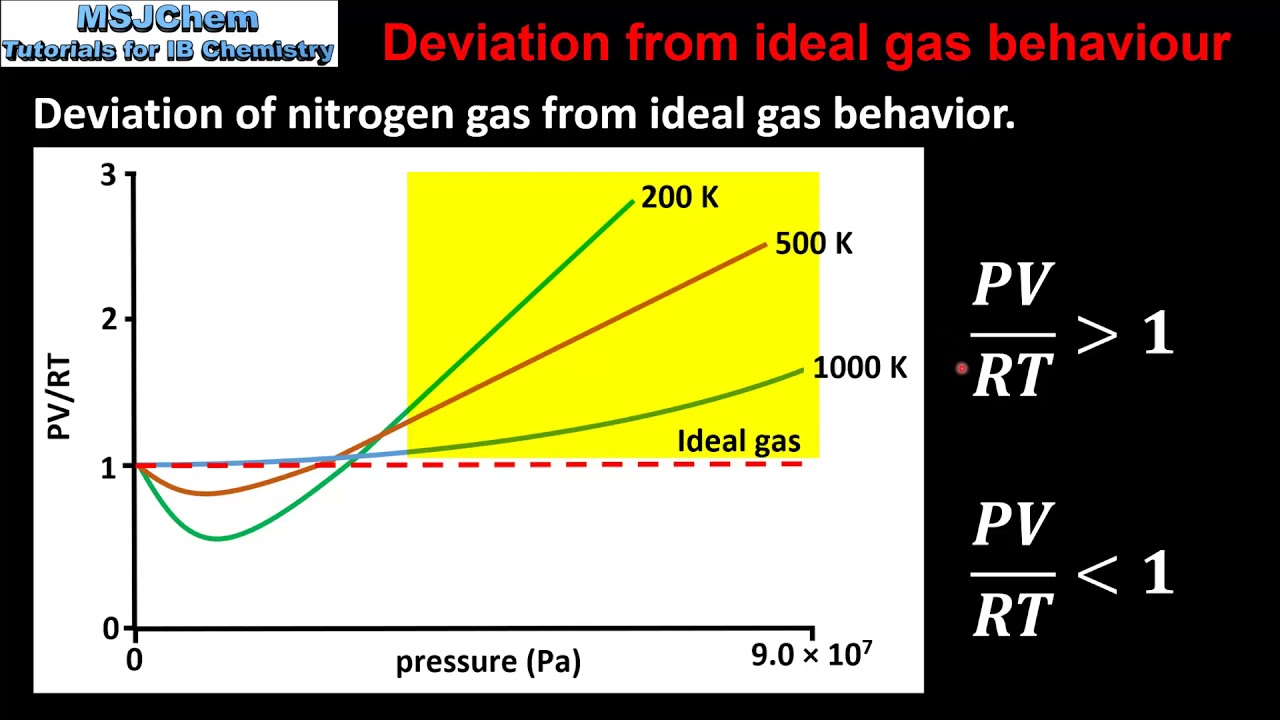
1 3 Deviation From Ideal Gas Behaviour YouTube

Chemistry Net Gas Laws Ideal Gas Law Pv nrt Calculator Ideal Gas

Gas What Is The Ideal Gas Law

R Values Ideal Gas Law Bar Gas Gas Law Constant It Is A Good

Pin By Jessica Joyce On Ideal Gas Law Ideal Gas Law Gas Laws

Ideal Gas Law Density wmv YouTube

https://chem.libretexts.org/Bookshelves/Physical_and_Theoretical_Chemistry_Textbook_Maps/Supplemental_Modules_(Physical_and_Theoretical_Chemistry)/Physical_Properties_of_Matter/States_of_Matter/Properties_of_Gases/Gas_Laws/The_Ideal_Gas_Law
The Ideal Gas Equation Before we look at the Ideal Gas Equation let us state the four gas variables and one constant for a better understanding The four gas variables are pressure P volume V number of mole of gas n and temperature T Lastly the constant in the equation shown below is R known as the the gas constant which will be discussed in depth further later
/GettyImages-1044456654-e456c93eeeaf46fe84cbeb0f95814fb6.jpg?w=186)
https://chem.libretexts.org/Courses/can/CHEM_210%3A_General_Chemistry_I%2C_An_%22Atoms_Up%22_Approach/15%3A_Gases_and_Gas_Laws/15.06%3A_Ideal_Gases_and_Real_Gases
At very high gas densities the classical gas laws can be very poor approximations As we have noted they are better approximations the lower the density of the gas In fact experiments show that the pressure volume temperature behavior of any real gasreal gas becomes arbitrarily close to that predicted by the ideal gas equation in the

https://www.khanacademy.org/science/physics/thermodynamics/temp-kinetic-theory-ideal-gas-law/a/what-is-the-ideal-gas-law
What is an elastic collision Ideal gas molecules themselves take up no volume The gas takes up volume since the molecules expand into a large region of space but the Ideal gas molecules are approximated as point particles that have no volume in and of themselves If this sounds too ideal to be true you re right

https://www.khanacademy.org/science/ap-chemistry-beta/x2eef969c74e0d802:intermolecular-forces-and-properties/x2eef969c74e0d802:ideal-gas-law/v/ideal-gas-equation-pv-nrt
About Transcript The ideal gas law PV nRT relates the macroscopic properties of ideal gases An ideal gas is a gas in which the particles a do not attract or repel one another and b take up no space have no volume No gas is truly ideal but the ideal gas law does provide a good approximation of real gas behavior under many conditions

https://chem.libretexts.org/Courses/University_of_Florida/CHM2047%3A_One-Semester_General_Chemistry_(Kleiman)/09%3A_Gases/9.02%3A_The_Ideal_Gas_Equation
The ideal gas law is derived from empirical relationships among the pressure the volume the temperature and the number of moles of a gas it can be used to calculate any of the four properties if the other three are known Ideal gas equation PV nRT where R 0 08206L atm K mol 8 3145 J K mol
The Ideal Gas Law may be expressed as PV NkT where P absolute pressure in atmospheres V volume usually in liters n number of particles of gas k Boltzmann s constant 1 38 10 23 J K 1 T temperature in Kelvin The ideal gas law also called the general gas equation is the equation of state of a hypothetical ideal gas It is a good approximation of the behavior of many gases under many conditions although it has several limitations
In such a case all gases obey an equation of state known as the ideal gas law PV nRT where n is the number of moles of the gas and R is the universal or perfect gas constant 8 31446261815324 joules per kelvin per mole The universal gas constant is defined as Avogadro s number NA times the Boltzmann constant k